Change of the peripheral blood immune pattern and its correlation with prognosis in patients with liver cancer treated by sorafenib
Chang-Jiang Lei, Jia-Ni Liu, Rong Wu, Zhi-Xiong Long, Jiang-Zhou Zhang, Dan Tao,Yan-Ping Liu1Tumor Laboratory, the Second Affiliated Hospital of Jianghan University, Wuhan, 40050, Hubei, ChinaGeriatric Department, the Central Hospital of Wuhan, Wuhan, 40014, Hubei, ChinaOncology Department, the Second Affiliated Hospital of Jianghan University, Wuhan, 40050, Hubei, China
Change of the peripheral blood immune pattern and its correlation with prognosis in patients with liver cancer treated by sorafenib
Chang-Jiang Lei1#, Jia-Ni Liu2#, Rong Wu1#, Zhi-Xiong Long3*, Jiang-Zhou Zhang3, Dan Tao3,Yan-Ping Liu31Tumor Laboratory, the Second Affiliated Hospital of Jianghan University, Wuhan, 430050, Hubei, China
2Geriatric Department, the Central Hospital of Wuhan, Wuhan, 430014, Hubei, China
3Oncology Department, the Second Affiliated Hospital of Jianghan University, Wuhan, 430050, Hubei, China
ARTICLE INFO ABSTRACT
Article history:
in revised form 16 March 2016 Accepted 15 April 2016
Available online 20 June 2016
Liver cancer
Sorafenib
Chemotherapy
Regulatory B cells Prognosis
Objective: To study the change of the peripheral blood immune pattern and its correlation with prognosis in patients with liver cancer after treated by sorafenib. Methods: Patients with advanced liver cancer admitted in our hospital were enrolled and treated with sorafenib. After two months of the treatment, their peripheral blood was collected. The immune cell subset and cytokines level were determined by flow cytometry and luminex technology. According to the reaction expressed by patients towards sorafenib, patients were divided into the response group and the no response group. The changes of the peripheral blood immune pattern and its correlation with prognosis of patients in the two groups were compared. Results: Before and after treatment of sorafenib, there was no significant difference in the ratios of T cells,NK cells and their subtypes in peripheral blood of patients between the two groups; while after treatment the ratio of B cells and regulatory B cells (Breg) of patients in the response group was significant higher than that of the no response group (P<0.05), and the prognosis conditions of patients with decreased ratio of Breg cells were better than other patients after undergoing chemotherapy. The levels of plasma cytokines IL-6, IL-10, IL-12, IL17, FIL-3L,IFN-γ, TNF-α, MCP-1 and VEGF showed no significant differences. Conclusions: After treatment of sorafenib, the prognosis conditions of patients of advanced liver cancer with a reduced Breg ratio are better than patients with an unaltered or increased Breg ratio. The ratio of Breg in peripheral blood may be considered as early biological indicator for the prediction of the curative effects of sorafenib.
1. Introduction
Liver cancer is one of the commonest malignant tumors in our country with a appropriate incidence rate of 1/10 000 and more than 600 000 new added cases each year in the world, which has a strong impact on mankind's health and life quality [1,2]. The detection rate of early liver cancer is about 30%-40% [3]. The majority of advanced liver cancer patients are not suitable for radical resection and liver transplantation. In short of effective treatment measures, patients live with a short survival period and poor prognosis [4-6]. Sorafenib is the first first-line drug which was approved to be applied in the systematic treatment of liver cancer. Many domestic and abroad clinical researches have proved that sorafenib can prolong the survival period of patients with advanced liver cancer and slow the development of cancer [3, 7-10]. However, sorafenib do not work for all liver cancer patients [11]. The reason remains to be further studied. Multiple studies reveal that chemotherapy can influence the immune system of cancer patients [12-14]. Then, dose the change of the immune pattern influence the effect of chemotherapy? This study aimed to explore the change of the immune pattern and its correlation with prognosis in patients with advanced liver cancer after treated by sorafenib by testing their immune patterns.
2. Materials and methods
2.1. Materials
Lymphocyte separation medium with a concentration of 1.077 used in this study was purchased from Sinopharm Chemical Group. Flow antibodies, cell fixed liquid and transmembrane liquid, such as human CD3 (marked by FITC), CD4 (marked by PE), CD8(marked by PB), CD56 (marked by APC), CD25 (marked by APCCy7), CD27 (marked by APC-Cy7), CD19 (marked by PE), CD24(marked by PE) and Foxp3 (marked by APC) were all bought from eBioscience Company. Human cytokines, Milliplex kits and Luminex 200 detection system were all from Millipore.
2.2. Methods
2.2.1. Clinical materials
A total of 73 advanced liver cancer patients who were treated in our hospital from September 2013 to March 2015 were selected and treated with sorafenib. Among them, five quitted the study due to severe adverse reactions, four cases did not return for further consultation and peripheral blood collection at the fourth week after using drugs, two lost to follow-up. Therefore a total of 62 cases were included in statistical analysis of this study. Among those included patients, 41 cases were males and 21 cases were females;24 cases were under 45 years old and 38 cases were over 45 years;18 cases underwent surgical treatment while 44 cases did not;37 cases suffered from metastasis, while 25 cases did not; alpha fetoprotein (AFP) was less than 50 in 14 cases; while it was more than 50 in 44 cases, 9 of which were caused by hepatitis C, 13 by excessive drinking, 12 by hepatitis B and the cause of the other 28 cases remained unknown. All patients were diagnosed as advanced liver cancer by pathology examination (could not accept surgical treatment or reoccurred after surgical and local treatment) with good liver, renal and hematopoietic functions. Patients received systemic anti-cancer treatment and patients with liver, renal and hematopoietic dysfunctions were excluded. All patients signed an informed consent. The study was approved by the Ethical Inspection Committee of our hospital.
2.2.2. Chemotherapy scheme and therapeutic effect evaluation
Patients met the inclusion criteria were treated with sorafenib orally 400 mg a time once every two days. Four cases showed severe side effects (hand and foot skin reaction, lipsotrichia, rash, diarrhea and hypertension) which did not involved in statistics. Therapeutic effect evaluation was conducted every three weeks after using drugs. The evaluation standard consulted the Response Evaluation Criteria in Solid Tumors (RECIST) [15].
2.2.3. Flow cytometry
Anticoagulant tube was used to collecte 5 mL peripheral blood of advanced liver cancer patients before and 4 weeks after treatment,respectively. Peripheral blood mononuclear cells (PBMC) were isolated by lymphocytes separating solution and then the upper serum was collected and stored at -80 ℃. PBMC were suspended again after washed by PBS with a regulated concentration of 1×106/mL. After blocked, flow antibodies were added for dying. And it was incubated in a cool dark place at 4 ℃ for 30 min and then detected after washed by PBS twice. Intracellular dyed cells incubated antibodies after treated by fixed transmembrane liquid. They were tested after washing by transmembrane liquid. The flow results were determined by BD FACS Calibur and analyzed by FlowJo software. Isotype control antibody-dyed cells were used as the negative control in the analysis of flow results. Surface markers such as CD3, CD4,CD8, CD25/Foxp3 and CD45RA/CD45RO were used to illustrate T cell subset, CD19 and CD24/CD25 were to analyze B cells, and CD56 was to mark NK cells.
2.2.4. Luminex
Plasma collected from patients with liver cancer was stored in the refrigerator at -80 and was not thawed and refrozen before detection. Milliplex kits and the corresponding Luminex 200 liquidchip system (Millipore) were applied to detect the levels of IL-6, IL-10, IL-12, IL17, FIL-3L, IFN-γ, TNF-α, MCP-1 and VEGF of plasma in patients. The operation procedure was strictly followed the constructions. Firstly, 25 μL diluent, 25 μL plasma and 25 μL mierospheres were mixed and added onto 96-well plates for incubation overnight. Then, after washed, 25 μL detected antibodies were added and incubated for 30 min. After that, 25 μL streptavidinphycoerythrin was added in after rewashed and again incubated for 30 min. Next, the plate was washed twice. Mierospheres were suspended on 100 μL sheath fluid and detected and analyzed by Luminex 200. The concentration of each cytokine was counted on a basis of the mean fluorescence intensity (MFI).
2.2.5. Statistical analysis
All data in this study were counted and analyzed by Graphpad Prism 4.0. The comparison between immune cell subset and cytokines of patients before and after treatment were tested with t-test. Means between two groups were analyzed by Student’s t-test. Differences remained to be statistically significant when P<0.05. Measurement data were expressed by mean ± SD.
3. Results
3.1. Treatment outcomes
In this study, a total of 62 patients were included , of which 41 were males and 21 were females. A follow-up of these patients was conducted after they were treated by sorafenib and the last follow-up was done on January 2016. The median survival time was 10.2 months,median progression free survival time was 3.9 months, and 22 patients survived. After treatment, 4 cases showed slight side effects which was lessened after symptomatic treatment. The effective rate reached 71% after patient were treated by sorafenib. The illness of 44 patients were controlled (42 cases were in stable conditions and 2 cases were in remission). Those patients were regarded as the response group. The rest 20 patients showed no responses towards sorafenib, so they were included in the no response group.
3.2. The distribution of peripheral blood immune cell subset of patients before and after treatment
The ratios of peripheral blood T cells, B cells, NK cells and theirsubsets of advanced liver cancer patients before and after treated by sorafenib were detected. The ratios of total T cells, CD4+ T cells,regulatory T cells, memory T cells and NK cells in the two groups showed no differences; while the ratio of B cells and regulatory B cells (Breg) in PBMC of patients in the response group was higher than that of in the no response group (P<0.05) (Table 1).
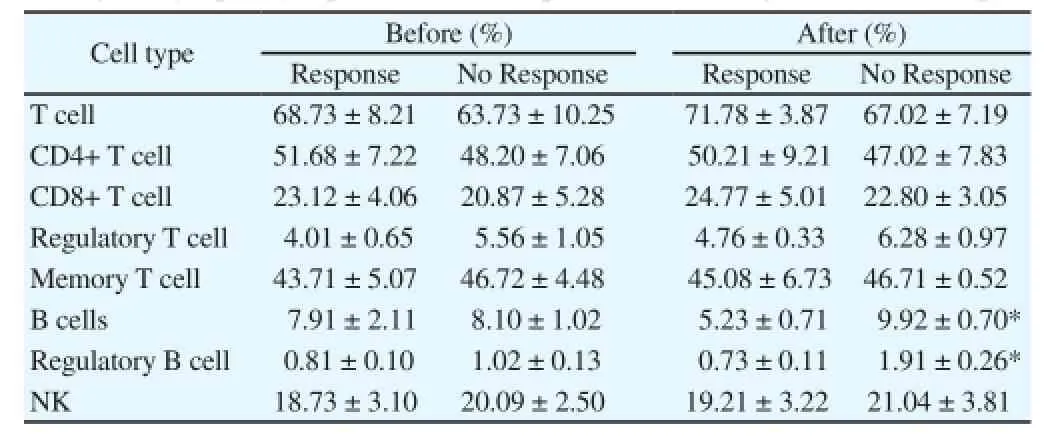
Table 1Change in lymphocyte profiles in HCC patients following sorafenib therapy.
3.3. The correlation of the ratio of Breg cells and prognosis of patients treated with chemotherapy
In order to further analyze the relationship between Breg cells and chemotherapy efficacy, according to the change of the Breg ratios in PBMC in patients before and after chemotherapy, we divided the subjects in to the Breg-reduced group (the ratio of Breg decreased more than half as compared with rpe-chemotherapy, n=29) and the no Breg-reduced group (n= 29). The median survival time of patients with increased or changeless Breg ratios after treaed with sorafenib was 9.3 (7.3-14.0) months, while the median survival time of patients with induced Breg ratios was 10.6 (9.0-18.2) months. Differences were statistically significant.
3.4. The level of cytokines in serum of patients before and after chemotherapy
Levels of multiple inflammatory cytokines including IL-6, IL-10,IL-12, IL17, FIL-3L, IFN-γ, TNF-α, MCP-1 and VEGF were tested simultaneously by luminex technology. The results showed that the level of cytokines in serum of patients in the response and no response groups had no statistical differences (Table 2).

Table 2The level of cytokines in serum of HCC patients following sorafenib therapy.
4. Discussion
Hepatocellular carcinoma is a clinically common cancer in our country with a pretty high malignant degree, strong wetting property and transitivity, high recurrence rate and poor prognosis. Patients dead from liver cancer each year in our country accounts for about 50% in the total number of the world, which has brought heavy burdens for our society [16]. At present, for patients with early liver cancer, surgical treatment of this disease is the first choice. However,it is difficult to diagnose the disease at the onset. Most patients have reached the locally advanced stage or gone through distant metastasis and therefore loss the best surgical time. Thus, the effective treatment of advance liver cancer is what we clinical workers are confronting. Sorafenib is the first effective molecular target drug in the world which was approved by FDA and proved to obviously prolong the survival time of HCC patients, and it is also recommended to be a first-line drug for HCC patients who should not be treated with surgery[16]. On the one hand, sorafenib can fight against cancer by inhibiting the activity of Raf kinase of cancer cells and interdicting the Ras/Raf/MAPK cascade signal pathway to inhibit the proliferation of cancer cells. On the other hand, sorafenib can also lower the activities of angiogenesis factors receptor -2 and -3 and plays a role of anti-angiogenesis [17, 18]. In this study, 62 cases with advanced liver cancer were treated with sorafenib. Illness of 44 of them has been controlled (42 cases were in stable conditions and 2 cases were in remission). The effective rate was 71% (44/62). The total medium survival time was 10.2 (7.3-18.2) months, which was matched with results of other domestic and abroad clinical researches [3, 19].
More and more evidences have clarified that the immune state of cancer patients is closely related to the growth and prognosis of tumors. Researchers have shown that there are many immune cells infiltrating the liver cancer tissues. Besides, the infiltration degree of FoxP3+ Treg and IL-17+ T cells was negatively correlated with the prognosis of patients [20]. Tumor-related macrophages can promote epithelial and stromal transformation and enhance the dryness of tumor cells by transforming growth factors [21]. Tumorrelated neutrophils can recruit Treg to the local tumor to facilitate the growth of tumor and strengthen its drug resistance [22]. However,due to the limitation of the specificity of samples, the infiltration condition of local tumor immune cells can provide no basis for the treatment of the disease. Therefore, in this study those advanced liver cancer patients were divided into the response and no response groups in accordance with the reactions they had shown after treated by sorafenib to analyze the types of peripheral blood immune cells and the cytokine level after they were treated for a month, which aimed to provide early indexes for the evaluation of prognosis for patients with chemotherapy and those patients to choose a suitable therapeutic regimen. It was found that before and after treated by sorafenib the ratio of T cells and NK cells of peripheral blood of the two groups had no differences, but the ratio of B cells and Breg cells of patients in the response group was significantly lower than that in the no response group (P < 0.05); while the serum cytokines (IL-6,IL-10, IL-12, IL17, FIL-3L, IFN-γ, TNF-α, MCP-1 and VEGF)of the two groups remained changeless, which indicated that the levels of these cytokines had little to do with the development and prognosis of the illness conditions of advanced liver cancer patients. That is to say they cannot serve as prediction indexes for the effect of sorafenib in patients with advanced liver cancer.As always, it is considered that B cells play immunologic functions to accelerate inflammatory reactions by presenting antibodies,secreting cytokines and mediating humoral immune responses[23, 24]. Recent studies have found that there is a series of Breg existing in bodies. Breg is an important B cell subset which could control immune responses by secreting IL-10 and transforming growth factor-β1 and other suppressive cytokines [25, 26]. There is a research manifesting that the ratio of Breg in local tumor area of liver cancer patients was higher than those in the tumor or non-tumor areas; the ratio of peripheral Breg of liver cancer patients was also higher than that of health people [27]. The ratio was closely related to neoplasm staging, tumor diversity and venous invasion. The research pointed out that Breg could promote the growth and infiltration of liver cancer cells by CD40/CD154 signal pathway. Our study results also discovered that after treated with sorafenib, patients with decreased Breg cells ratio lived longer than those patients whose Breg cell ratio increased. The above research results showed that the ratio of peripheral blood Breg cells in advanced liver cancer patients is closely related to the development and prognosis of the disease. It can therefore serve as early biological indicator for the prediction of the curative effect of chemotherapy so as to provide references for the optimization of the therapeutic regimen.
Declare of interest statement
We declare that we have no conflict of interest.
References
[1] Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, et al. Cancer statistics in China, 2015. CA Cancer J Clin 2016; 66(2): 115-132.
[2] Zhou M, Wang H, Zhu J, Chen W, Wang L, Liu S, et al. Cause-specific mortality for 240 causes in China during 1990-2013: a systematic subnational analysis for the global burden of disease study 2013. Lancet 2016; 387(10015): 251-272.
[3] Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med 2008;359(4): 378-390.
[4] Giannini EG, Farinati F, Ciccarese F, Pecorelli A, Rapaccini GL, Di Marco M, et al. Prognosis of untreated hepatocellular carcinoma. Hepatology 2015; 61(1): 184-190.
[5] Mazzanti R, Arena U, Tassi R. Hepatocellular carcinoma: Where are we?World J Exp Med 2016; 6(1): 21-36.
[6] Forner A, Llovet JM, Bruix J. Hepatocellular carcinoma. Lancet 2012;379(9822): 1245-1255.
[7] Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, et al. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebocontrolled trial. Lancet Oncol 2009; 10(1): 25-34.
[8] Kudo M, Imanaka K, Chida N, Nakachi K, Tak WY, Takayama T, et al. Phase III study of sorafenib after transarterial chemoembolisation in Japanese and Korean patients with unresectable hepatocellular carcinoma. Eur J Cancer 2011; 47(14): 2117-2127.
[9] Bai W, Wang YJ, Zhao Y, Qi XS, Yin ZX, He CY, et al. Sorafenib in combination with transarterial chemoembolization improves the survival of patients with unresectable hepatocellular carcinoma: a propensity score matching study. J Dis Dis 2013; 14(4): 181-190.
[10] Zhu AX, Rosmorduc O, Evans TR, Ross PJ, Santoro A, Carrilho FJ, et al. SEARCH: a phase III, randomized, double-blind, placebo-controlled trial of sorafenib plus erlotinib in patients with advanced hepatocellular carcinoma. J Clin Oncol 2015; 33(6): 559-566.
[11] Granito A, Marinelli S, Negrini G, Menetti S, Benevento F, Bolondi L. Prognostic significance of adverse events in patients with hepatocellular carcinoma treated with sorafenib. Therap Adv Gastroenterol 2016; 9(2): 240-249.
[12] Yang J, Li W, Luo F, Zhao N, Zhang W, Zhang D, et al. Low percentage of CD24hiCD27(+)CD19(+) B cells decelerates gastric cancer progression in XELOX-treated patients. Int Immunopharmacol 2015;26(2): 322-327.
[13] Maskey N, Li K, Hu M, Xu Z, Peng C, Yu F, et al. Impact of neoadjuvant chemotherapy on lymphocytes and co-inhibitory B7-H4 molecule in gastric cancer: low B7-H4 expression associates with favorable prognosis. Tumour Biol 2014; 35(12): 11837-11843.
[14] Weir GM, Liwski RS, Mansour M. Immune modulation by chemotherapy or immunotherapy to enhance cancer vaccines. Cancers 2011; 3(3): 3114-3142.
[15] Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS,Rubinstein L, et al. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Nati Cancer Inst 2000; 92(3): 205-216.
[16] Bruix J, Takayama T, Mazzaferro V, Chau GY, Yang J, Kudo M, et al. Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): a phase 3, randomised, double-blind, placebocontrolled trial. Lancet Oncol 2015; 16(13): 1344-1354.
[17] Gao JJ, Shi ZY, Xia JF, Inagaki Y, Tang W. Sorafenib-based combined molecule targeting in treatment of hepatocellular carcinoma. World J Gastroenterol 2015; 21(42): 12059-12070.
[18] Nishida N, Kitano M, Sakurai T, Kudo M. Molecular mechanism and prediction of sorafenib chemoresistance in human hepatocellular carcinoma. Dig Dis 2015; 33(6): 771-779.
[19] Al-Rajabi R, Patel S, Ketchum NS, Jaime NA, Lu TW, Pollock BH, et al. Comparative dosing and efficacy of sorafenib in hepatocellular cancer patients with varying liver dysfunction. J Gastrointest Oncol 2015; 6(3): 259-267.
[20] Huang Y, Wang F, Wang Y, Zhu Z, Gao Y, Ma Z, Xu R, et al. Intrahepatic interleukin-17+ T cells and FoxP3+ regulatory T cells cooperate to promote development and affect the prognosis of hepatocellular carcinoma. J Gastroenterol Hepatol 2014; 29(4): 851-859.
[21] Fan QM, Jing YY, Yu GF, Kou XR, Ye F, Gao L, et al. Tumor-associated macrophages promote cancer stem cell-like properties via transforming growth factor-beta1-induced epithelial-mesenchymal transition in hepatocellular carcinoma. Cancer Lett 2014; 352(2): 160-168.
[22] Zhou SL, Zhou ZJ, Hu ZQ, Huang XW, Wang Z, Chen EB, et al. Tumorassociated neutrophils recruit macrophages and t-regulatory cells to promote progression of hepatocellular carcinoma and resistance to sorafenib. Gastroenterology 2016; doi:10.1053/j.gastro.2016.02.040.
[23] Igarashi K, Itoh-Nakadai A. Orchestration of B lymphoid cells and their inner myeloid by Bach. Curr Opin Immunol 2016; 39:136-142.
[24] Shen P, Fillatreau S. Antibody-independent functions of B cells: a focus on cytokines. Nat Rev Immunol 2015; 15(7): 441-451.
[25] Rosser EC, Mauri C. Regulatory B cells: origin, phenotype, and function. Immunity 2015; 42(4): 607-612.
[26] Mauri C, Blair PA. The incognito journey of a regulatory B cell. Immunity 2014; 41(6): 878-880.
[27] Shao Y, Lo CM, Ling CC, Liu XB, Ng KT, Chu AC, et al. Regulatory B cells accelerate hepatocellular carcinoma progression via CD40/CD154 signaling pathway. Cancer Lett 2014; 355(2): 264-272.
Document heading 10.1016/j.apjtm.2016.04.019
15 February 2016
#These authors contributed equally to this work.
*Corresponding author: Zhi-Xiong Long, Geriatrics, the central hospital of wuhan,Wuhan, 430050, Hubei, China.
Tel: 18271483056
E-mail:hkfc98@tom.com
Foundation project: This study was supported by the Applied Basic Research Project of Wuhan Municipal Science and Technology Bureau (Grant No. 2015061701011630,NO.2013062301010823), Medical Scientific Research project of the Education Department of Hubei Province (Grant No. B2015230) and the fourth batch of Hanyang Concert Talent Plan.
 Asian Pacific Journal of Tropical Medicine2016年6期
Asian Pacific Journal of Tropical Medicine2016年6期
- Asian Pacific Journal of Tropical Medicine的其它文章
- Experiment research of cisplatin implants inhibiting transplantation tumor growth and regulating the expression of KLK7 and E-cad of tumor-bearing mice with gastric cancer
- Study on prevention effect of Zishen Yutai pill combined with progesterone for threatened abortion in rats
- Correlation study of biological characteristics of non-small cell lung cancer A549 cells after transfecting plasmid by microbubble ultrasound contrast agent
- Expression and significance of angiostatin, vascular endothelial growth factor and matrix metalloproteinase-9 in brain tissue of diabetic rats with ischemia reperfusion
- Comparative study of the chitooligosaccharides effect on the proliferation inhibition and radiosensitization of three types of human gastric cancer cell line
- Are efforts up to the mark? A cirrhotic state and knowledge about HCV prevalence in general population of Pakistan
