川芎嗪注射液、α-酮酸联合左旋氨氯地平治疗慢性肾功能衰竭的临床观察
杨雪莲,李列平(凉山彝族自治州第二人民医院,四川西昌 615000)
川芎嗪注射液、α-酮酸联合左旋氨氯地平治疗慢性肾功能衰竭的临床观察
杨雪莲*,李列平(凉山彝族自治州第二人民医院,四川西昌615000)
目的:观察川芎嗪注射液、α-酮酸联合左旋氨氯地平治疗慢性肾功能衰竭的疗效和安全性。方法:92例慢性肾功能衰竭患者随机分为观察组(46例)和对照组(46例)。所有患者均给予电解质、维生素及氨基酸、低盐低脂、降糖降脂、抗感染及纠正贫血等常规对症治疗。在此基础上,对照组患者口服复方α-酮酸片8片,每日3次+苯磺酸左旋氨氯地平片1片,每日1次。观察组患者在对照组治疗的基础上给予盐酸川穹嗪注射液150 mg,加入5%葡萄糖250 ml中,静脉滴注,每日1次。两组疗程均为3个月。观察两组患者的临床疗效,治疗前后尿素氮(BUN)、血肌酐(Scr)、血清胱抑素(CysC)、低密度脂蛋白胆固醇(LDL-C)、总胆固醇(TC)、三酰甘油(TG)水平及不良反应发生情况。结果:观察组患者总有效率显著高于对照组,差异有统计学意义(P<0.05)。治疗前,两组患者BUN、Scr、CysC、LDL-C、TC、TG比较,差异均无统计学意义(P>0.05)。治疗后,两组患者BUN、Scr、LDL-C、TC、TG均显著低于同组治疗前,且观察组低于对照组,CysC显著高于同组治疗前,且观察组高于对照组,差异均有统计学意义(P<0.05)。两组患者不良反应发生率比较,差异无统计学意义(P>0.05)。结论:在常规治疗的基础上,川芎嗪注射液、α-酮酸联合左旋氨氯地平治疗慢性肾功能衰竭的疗效显著,可改善肾功能,且安全性较好。
川芎嗪注射液;α-酮酸;左旋氨氯地平;慢性肾功能衰竭;疗效;安全性
慢性肾功能衰竭(CRF)是一种严重危害患者生命健康的常见疾病,虽然其进展过程较为缓慢,但该疾病的病程一般难以逆转[1-2]。目前,对于CRF的治疗,临床多采用透析及肾脏移植等方法,尽管有较好的疗效,但价格较昂贵,患者经济上无力承受,导致无法普及,因此寻找有效、经济的治疗CRF的方法就显得极为重要[3]。有报道显示,左旋氨氯地平联合α-酮酸治疗CRF能够显著提高疗效,改善患者临床症状[4]。川芎嗪是现代化中药制剂,由于其具有较好的抑制血小板聚集作用,因而成为治疗CRF的非透析药物之一[5]。为此,在本研究中笔者观察了川芎嗪注射液、α-酮酸联合左旋氨氯地平治疗CRF的疗效和安全性,旨在为临床提供参考。
1 资料与方法
1.1研究对象
选择2012年4月-2015年4月我院肾内科接受治疗的92 例CRF患者,其中男性42例,女性50例;年龄21~65岁,平均年龄(41.2±5.1)岁;病程2~16年,平均病程(8.7±0.6)年;疾病种类:糖尿病肾病17例,慢性肾小球肾炎49例,多囊肾20例,高血压性肾小球动脉硬化6例。按随机数字表法将所有患者分为观察组(46例)和对照组(46例)。两组患者性别、年龄、病程等基本资料比较,差异均无统计学意义(P>0.05),具有可比性,详见表1。本研究方案经医院医学伦理委员会审核通过,所有患者或其家属均签署了知情同意书。
表1 两组患者基本资料比较Tab 1 Comparison of basic information between 2 groups

表1 两组患者基本资料比较Tab 1 Comparison of basic information between 2 groups
组别观察组对照组n 糖尿病肾病,例多囊肾,高血压性肾小球动脉硬化,例46 46年龄,岁41.0±5.0 41.5±5.3男性/女性,例20/26 22/24病程,年8.4±0.5 8.9±0.7 89慢性肾小球肾炎,例25 24 例91 1 42
1.2纳入与排除标准
纳入标准:(1)均符合CRF的相关诊断标准[6],且为非终末期患者;(2)血肌酐(Scr)178~707 μmol/L或内生肌酐清除率(Ccr)10~50 ml/min。排除标准:(1)同时患有其他严重系统性疾病,如肝癌等恶性肿瘤者;(2)严重的心血管疾病及精神病患者;(3)妊娠期和哺乳期妇女;(4)对药物过敏或过敏体质者。
1.3治疗方法[7]
所有患者均给予电解质、维生素及氨基酸、低盐低脂、降糖降脂、抗感染及纠正贫血等常规对症治疗。在此基础上,对照组患者口服复方α-酮酸片(北京费森尤斯卡比医药有限公司,规格:0.63 g/片,批准文号:国药准字H20041442)8片,每日3次+苯磺酸左旋氨氯地平片(施慧达药业集团有限公司,规格:2.5 mg/片,批准文号:国药准字H19991083)1片,每日1次。观察组患者在对照组治疗的基础上给予盐酸川穹嗪注射液(北京市燕京药业集团,规格:2 ml∶40 mg,批准文号:国药准字H20043160)150 mg,加入5%葡萄糖250 ml中,静脉滴注,每日1次。两组疗程均为3个月。
1.4观察指标
观察两组患者治疗前后尿素氮(BUN)、Scr、血清胱抑素(CysC)、低密度脂蛋白胆固醇(LDL-C)、总胆固醇(TC)、三酰甘油(TG)水平及不良反应发生情况。
1.5疗效判定标准[8]
显效:临床症状减少>80%,肾功能指标明显改善;有效:临床症状减少50%~80%,肾功能指标有所改善;无效:临床症状、肾功能指标均无改善或加重。总有效率=(显效例数+有效例数)/总例数×100%。
1.6统计学方法
采用SPSS 22.0统计软件对数据进行分析。计量资料以表示,采用t检验;计数资料以%表示,采用χ2检验。P<0.05为差异有统计学意义。
2 结果
2.1两组患者临床疗效比较
观察组患者总有效率显著高于对照组,差异有统计学意义(P<0.05),详见表2。

表2 两组患者临床疗效比较[例(%)]Tab 2 Comparison of clinic efficacy between 2 groups[case (%)]
2.2两组患者治疗前后BUN、Scr、CysC水平比较
治疗前,两组患者BUN、Scr、CysC水平比较,差异均无统计学意义(P>0.05)。治疗后,两组患者BUN、Scr水平均显著低于同组治疗前,且观察组低于对照组,CysC水平显著高于同组治疗前,且观察组高于对照组,差异均有统计学意义(P<0.05),详见表3。
表3 两组患者治疗前后BUN、Scr、CysC水平比较Tab 3 Comparison of BUN,Scr and CysC levels between 2groups before and after treatment

表3 两组患者治疗前后BUN、Scr、CysC水平比较Tab 3 Comparison of BUN,Scr and CysC levels between 2groups before and after treatment
注:与治疗前比较,*P<0.05;与对照组比较,#P<0.05Note:vs.before treatment,*P<0.05;vs.control group,#P<0.05
组别观察组对照组n治疗后36.2±3.5*#29.5±3.1*46 46 BUN,mmol/L治疗前21.1±2.1 21.3±2.2治疗后13.9±1.4*#17.2±1.7*Scr,mmol/L治疗前341.9±31.2 337.2±30.7治疗后142.4±21.0*#231.3±26.2*CysC,ml/min治疗前21.7±2.2 21.2±1.9
2.3两组患者治疗前后LDL-C、TC、TG水平比较
治疗前,两组患者LDL-C、TC、TG水平比较,差异均无统计学意义(P>0.05)。治疗后,两组患者LDL-C、TC、TG水平均显著低于同组治疗前,且观察组低于对照组,差异均有统计学意义(P<0.05),详见表4。
表4 两组患者治疗前后LDL-C、TC、TG水平比较,mmol/L)Tab 4 Comparison of LDL-C,TC and TG levels between 2 groups before and after treatment,mmol/L)
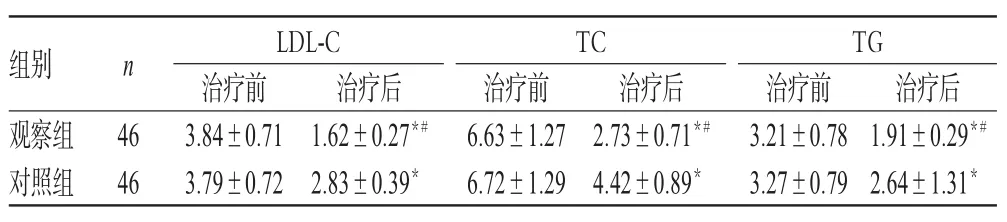
表4 两组患者治疗前后LDL-C、TC、TG水平比较,mmol/L)Tab 4 Comparison of LDL-C,TC and TG levels between 2 groups before and after treatment,mmol/L)
注:与治疗前比较,*P<0.05;与对照组比较,#P<0.05Note:vs.before treatment,*P<0.05;vs.control group,#P<0.05
治疗后1.91±0.29*#2.64±1.31*组别观察组对照组n 46 46 LDL-C治疗前3.84±0.71 3.79±0.72治疗后1.62±0.27*#2.83±0.39*TC治疗前6.63±1.27 6.72±1.29治疗后2.73±0.71*#4.42±0.89*TG治疗前3.21±0.78 3.27±0.79
2.4不良反应
观察组患者出现2例恶心、4例皮疹,不良反应发生率为13.0%;对照组患者出现2例恶心、3例皮疹,不良反应发生率为10.8%。两组患者不良反应发生率比较,差异无统计学意义(P>0.05)。治疗期间所有患者均未见严重不良反应发生。
3 讨论
CRF是肾功能不全的严重阶段,是由于各种原因造成人体出现慢性进行性的肾实质损害,最终可导致肾脏明显萎缩,无法维持器官的基本功能,出现电解质及酸碱平衡紊乱,全身各系统受累和机体代谢产物潴留等临床综合征,亦称为尿毒症。合理的药物治疗能够延缓疾病进展,少数患者可完全逆转病情,因此选择有效的药物治疗CRF显得至关重要[9-10]。α-酮酸可用于治疗由于慢性肾功能不全而引起的蛋白质代谢失调[11-12]。左旋氨氯地平是一种钙离子拮抗药,不仅可降低血压及肾小球内压,还能够防止肾小球硬化,缓解肾病相关症状[13-14]。有研究显示,中西医结合治疗CRF较单一西药治疗效果更好[15]。川穹嗪注射液可用于多种心血管等疾病的治疗,近年来被广泛运用于治疗CRF并取得了较好的疗效[16]。
本研究结果显示,观察组患者总有效率显著高于对照组,差异有统计学意义。这提示,川穹嗪注射液、α-酮酸联合左旋氨氯地平可明显提高治疗效果。BUN、Scr及CysC是评价肾功能的重要指标,本研究分别对治疗期间上述指标的变化情况进行检测,通过对该变化的探讨来研究用药期间患者肾功能的改变情况。本研究结果显示,治疗后,两组患者BUN、Scr均显著低于同组治疗前,且观察组低于对照组,CysC显著高于同组治疗前,且观察组高于对照组,差异均有统计学意义。这说明,川穹嗪注射液、α-酮酸联合左旋氨氯地平可改善CRF患者的肾功能。血脂相关指标的含量在CRF中具有重要意义。本研究结果表明,治疗后,两组患者LDL-C、TC、TG均显著低于同组治疗前,且观察组低于对照组,差异均有统计学意义。该结果出现的原因可能与川穹嗪具有降低血脂的作用有关。
综上所述,在常规治疗的基础上,川芎嗪注射液、α-酮酸联合左旋氨氯地平治疗CRF的疗效显著,可改善肾功能,且安全性较好。由于本研究纳入的样本量较小,检测指标较少;故此结论有待大样本、多指标研究进一步证实。
[1]Chu L,Zhang K,Zhang Y,et al.Mechanism underlying an elevated serum bile acid level in chronic renal failure patients[J].Int Urol Nephrol,2015,47(2):345.
[2]Huang Z,He L,Huang D,et al.Icariin protects rats against 5/6 nephrectomy-induced chronic kidney failure by increasing the number of renal stem cells[J].BMC Complement Altern Med,2015,15(1):378.
[3]Tbahriti HF,Messaoudi A,Kaddous A,et al.The degree of chronic renal failure is associated with the rate of pro-inflammatory cytokines,hyperhomocysteinemia and with oxidative stress[J].Ann Cardiol Angeiol:Paris,2014,63(3):135.
[4]Lemos CC,Mandarim-de-Lacerda CA,Carvalho JJ,et al.Gender-related differences in kidney of rats with chronic renal failure[J].Histol Histopathol,2014,29(4):479.
[5]李中和,刘章锁,邢国兰.川芎嗪对增殖性肾炎病人血浆内皮素和脂质过氧化物的影响[J].中国新药与临床杂志,2002,21(4):239.
[6]何玉华,赵良斌,林小风,等.肾康注射液联合血液透析治疗慢性肾功能衰竭临床观察[J].辽宁中医药大学学报,2013,15(9):135.
[7]Gulsahi A,Evirgen S,Öztaş B,et al.Volatile sulphur compound levels and related factors in patients with chronic renal failure[J].J Clin Periodontol,2014,41(8):814.
[8]Verma DK,Thelekkat Y,Bansal S.Metastatic calcification of floor of the mouth secondary to chronic renal failure-report of a rare case with atypical presentation[J].Dentomaxillofac Radiol,2015,44(5):20 140 023.
[9]Reuter SB,Harutyunyan M,Mygind ND,et al.The influence of contrast media on kidney function in patients with stable coronary artery disease[J].Scand Cardiovasc J,2014,48(4):234.
[10]Saeed A,DiBona GF,Grimberg E,et al.High-NaCl diet impairs dynamic renal blood flow autoregulation in rats with adenine-inducedchronic renal failure[J].Am J Physiol Regul Integr Comp Physiol,2014,306(6):R411.
[11]Khan JN,Wilmot EG,Leggate M,et al.Subclinical diastolic dysfunction in young adults with Type 2 diabetes mellitus:a multiparametriccontrast-enhanced cardiovascular magnetic resonance pilot study assessing potential mechanisms[J].Eur Heart J Cardiovasc Imaging,2014,15(11):1 263.
[12]Thilly N.Low-protein diet in chronic kidney disease:from questions of effectiveness to those of feasibility[J]/Nephrol Dial Transplant,2013,28(9):2 203.
[13]Piccoli GB,Ferraresi M,Deagostini MC,et al.Vegetarian low-protein diets supplemented with keto analogues:a niche for the few or an option for many[J].Nephrol Dial Transplant,2013,28(9):2 295.
[14]Qi Y,Zhang X.Determination of enantiomeric impurity of levamlodipine besylate bulk drug by capillary electrophoresis using carboxymethyl-β-cyclodextrin[J].Cell Biochem Biophys,2014,70(3):1 633.
[15]Yang YL,Yu LT,Wu ZT,et al.Synergic effects of levamlodipine and bisoprolol on blood pressure reduction and organ protection in spontaneously hypertensive rats [J].CNS Neurosci Ther,2012,18(6):471.
[16]樊相军,白桂花.丹参川芎嗪注射液治疗慢性肾功能衰竭30例临床分析[J].山西医药杂志,2012,41(1):38.
(编辑:陈宏)
Clinical Observation of Ligustrazine Injection,α-ketoacid Combined with Levamlodipine in the Treatment of Chronic Renal Failure
YANG Xuelian,LI Lieping(The Second People’s Hospital of Liangshan Yi Autonomous Prefecture,Sichuan Xichang 615000,China)
OBJECTIVE:To observe the clinical efficacy and safety of Ligustrazine injection,α-ketoacid combined with levamlodipine in the treatment of chronic renal failure.METHODS:92 patients with chronic renal failure were randomly divided into observation group(46 cases)and control group(46 cases).All patients received electrolyte,vitamins and essential amino acid,low salt,low fat,hypoglycemic and hypolipemic,anti-infection,correcting anemia and other conventional symptomatic treatment/Based on it,control group received 8 Compound α-ketoacid tablets,3 times a day+1 Benzenesulfonic acid levamlodipine tablet,once a day.Observation group additionally received 150 mg Ligustrazine hydrochloride injection,adding into 250 ml 5%Glucose injection,intravenous infusion,once a day.The treatment course for both groups was 3 months.Clinical efficacy,and urea nitrogen(BUN),serum creatinine(Scr),serum cystatin(CysC),low-density lipoprotein cholesterol(LDL-C),total cholesterol (TC),triglyceride(TG)levels before and after treatment and the incidence of adverse reactions in 2 groups were observed.RESULTS:The total effective rate in observation group was significantly higher than control group,the difference was statistically significant(P<0.05).Before treatment,there were no significant differences in the BUN,Scr,CysC,LDL-C,TC and TG between 2 groups(P>0.05).After treatment,BUN,Scr,LDL-C,TC and TG in 2 groups were significantly lower than before,and observation group was lower than control group,CysC was significantly higher than before,and observation group was higher than control group,the differences were statistically significant(P<0.05).And there was no significant difference in the incidence of adverse reactions(P>0.05).CONCLUSIONS:Based on conventional treatment,Ligustrazine injection,α-ketoacid combined with levamlodipine show obvious efficacy in the treatment of chronic renal failure,they can improve the renal functions,with good safety.
Ligustrazine injection;α-ketoacid;Levamlodipine;Chronic renal failure;Efficacy;Safety
R453文献标志码A
1001-0408(2016)24-3371-03
10.6039/j.issn.1001-0408.2016.24.19
*副主任医师。研究方向:肾内科,肾脏疾病诊治及血液净化。E-mail:2664381970@qq.com
2015-12-10
2016-06-16)

